Heart
- Let's Biologue!
- Sep 28, 2021
- 3 min read
Updated: May 18, 2023

The heart is a muscular organ that varies in size from individual to individual (usually ie fist) and pumps blood through the body (cardiovascular system).
The heart lies in the pericardial sac (conical sac of fibrous tissue) which is divided into the fibrous pericardium, which provides support and protection for the heart, and the serous pericardium (visceral and perinatal).
Three layers of the heart
Epicardium - the outer lining of the cardiac chamber constituted by simple squamous epithelia;
Myocardium - intermediate layer contains important structures such as excitable nodal tissue and the conducting system, constituted by cardiac muscle cells;
Endocardium - the innermost layer of the heart is formed of the endothelium and subendothelial connective tissue;

Four chambers of the internal cavity
Right atrium - receives deoxygenated blood from systemic veins
Left atrium - the left atrium receives oxygenated blood from the pulmonary veins
Right ventricle - pumps blood for the lungs
Left ventricle - pumps blood for the body
To keep the blood flowing in the correct direction heart has valves. The valves between the atria de ventricles are the atrioventricular valves (cuspid valves) while those leaving the ventricles are semilunar valves.
Right atrioventricular valve - tricuspid valve
Left atrioventricular valve - mitral ou bicuspid valve
Right ventricle - pulmonary semilunar valve
Left ventricular - aortic semilunar valve

The heart works rhythmically when the ventricles contract the tricuspid and bicuspid valve close and prevents blood from flowing back. Ventricle relaxes and semilunar valves close to prevent blood from flowing back into the ventricles.
Hypertrophic Cardiomyopathy

Hypertrophic cardiomyopathy (HCM) is the most prevalent inherited heart disease.
It is clinically defined by the presence of unexplained left ventricular hypertrophy.
Histologically, it is characterized by a myocardial architecture with disarrangement of cardiomyocytes and myofibrils, an increased wall thickness of intramural coronary arteries, and interstitial fibrosis.
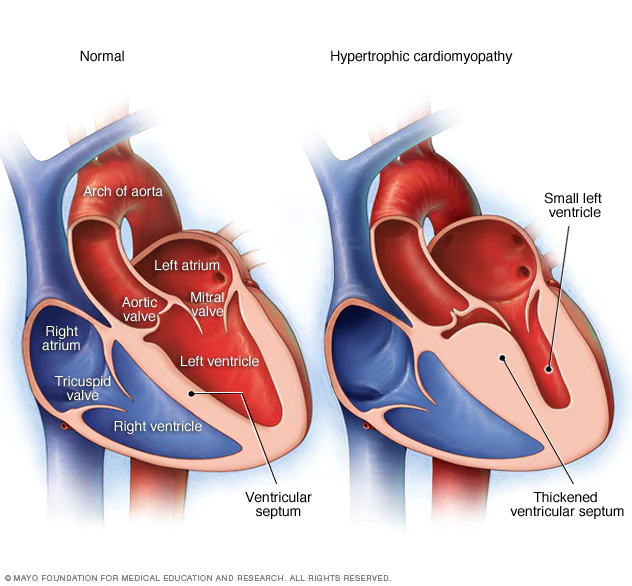
This disease is the leading cause of sudden death in individuals under 35 years of age, especially among young athletes.
There are more than 1,500 mutations in at least 11 different genes that have been identified as a potential cause for hypertrophic cardiomyopathy, which translates into great clinical heterogeneity, highlighting the need for individualized treatment approaches for each patient with HCM.
For this disease, there is no cure, only some therapies are described.
One of the most used therapeutic perspectives is diltiazem (a blocker of L-type Ca2+ channels indicated as a therapy for symptomatic relief since it is recognized that mutations in sarcomere genes associated with HCM lead to an early dysregulation of intracellular Ca2+ homeostasis).
Case study
Prondzynski and his collaborators, published in 2019, an article in which they intended to elucidate the functional role of a new mutation in ACTN2.
ACTN2 codes for α-actinin 2, which is a protein located in the Z disk of the sarcomere (a basic component of the striated muscle that allows muscle contraction) and its main function is the formation of sarcomeres.
For this, they studied the functional and contractile differences of cardiomyocytes mutated in ACTN2 with normal cardiomyocytes, all studies performed in vitro.

Through this study, they concluded that mutated cardiomyocytes, compared to controls (normal cardiomyocytes), had:
myofibrillar disarray
hypercontractility
prolonged relaxation
higher myofilament Ca2+ sensitivity
Based on this, this group of researchers experimented with the use of diltiazem as a potential treatment.
The results of this study demonstrated that diltiazem improved cardiomyocyte structure and contraction.
References
Elliott, P., & McKenna, W. J. (2004). Hypertrophic cardiomyopathy. Lancet, 363(9424), 1881–1891. https://doi.org/10.1016/S0140-6736(04)16358-7
Junqueira, L. C. U., & Carneiro, J. (2013). Histologia basic.
Maltês, S., & Lopes, L. R. (2020). New perspectives in the pharmacological treatment of hypertrophic cardiomyopathy. Revista Portuguesa de Cardiologia, xx. https://doi.org/10.1016/j.repc.2019.03.008
Prondzynski, M., Lemoine, M. D., Zech, A. T., Horváth, A., Di Mauro, V., Koivumäki, J. T., Kresin, N., Busch, J., Krause, T., Krämer, E., Schlossarek, S., Spohn, M., Friedrich, F. W., Münch, J., Laufer, S. D., Redwood, C., Volk, A. E., Hansen, A., Mearini, G., … Carrier, L. (2019). Disease modeling of a mutation in α‐actinin 2 guides clinical therapy in hypertrophic cardiomyopathy. EMBO Molecular Medicine, 11(12), 1–18. https://doi.org/10.15252/emmm.201911115
Prondzynski, M., Mearini, G., & Carrier, L. (2019). Gene therapy strategies in the treatment of hypertrophic cardiomyopathy. Pflugers Archiv European Journal of Physiology, 471(5), 807–815. https://doi.org/10.1007/s00424-018-2173-5
SEER Training Modules, Structure of the Heart. U. S. National Institutes of Health, National Cancer Institute. 20/08/2021 <https://training.seer.cancer.gov/>.
Shah S., Gnanasegaran G., Sundberg-Cohon J., Buscombe J. (2009) The Heart: Anatomy, Physiology and Exercise Physiology. In: Movahed A., Gnanasegaran G., Buscombe J., Hall M. (eds) Integrating Cardiology for Nuclear Medicine Physicians. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-540-78674-0_1
Standring, S., MBE, PhD, DSc, FKC, FAS, H., & FRCS, H. (Eds.). (2016). Gray´s Anatomy The Anatomical Basis of Clinical practice (fourty-first). Elsevier.
Weinhaus A.J., Roberts K.P. (2005) Anatomy of the Human Heart. In: Iaizzo P.A. (eds) Handbook of Cardiac Anatomy, Physiology, and Devices. Humana Press. https://doi.org/10.1007/978-1-59259-835-9_4
https://www.heartfoundation.org.nz/your-heart/heart-conditions/heart-valve-disease
https://www.physio-pedia.com/Anatomy_of_the_Human_Heart
https://www.webmd.com/heart/picture-of-the-heart
https://www.liverpool.ac.uk/~trh/local_html/heartdisease/pericardium.htm
.png)



Comments